Core Technology
HHP (High Hydrostatic Pressure) Decellularization
A physical method that maximizes native ECM biological integrity — not a chemical process that compromises it
Why It Matters
HHP's Essence: Preserve, Not Compromise
High Hydrostatic Pressure (HHP) decellularization applies uniform isostatic pressure of 600–1000 MPa to precisely disrupt cell membranes while producing minimal perturbation to the extracellular matrix's three-dimensional protein network, growth factors, and ultrastructure. Unlike chemical methods (SDS), which rely on detergents, HHP achieves decellularization through pure physical force — no chemical residues, no chemical degradation risk, ~91% bioactive component retention. This means the ECM partners receive has bioactivity approaching that of native tissue.
From Source Tissue to High-Bioactivity ECM: Five-Step Process
01
Tissue Harvesting & Preparation
Qualified source tissues selected and pre-processed to rigorous quality standards
02
HHP Treatment — Precise Decellularization, Full ECM Preservation
Tissues undergo isostatic treatment at 600–1000 MPa. Pressure uniformly applied in all directions precisely disrupts cell membranes while ECM protein networks and ultrastructure remain fully intact — with no chemical agents involved
03
Washing & Purification — Remove Immunogens, Preserve Bioactivity
Thorough removal of cellular debris, DNA fragments, and immunogenic components while maximally preserving growth factors, matrix proteins, and bioactive signaling molecules within the ECM
04
Quality Verification
Each batch verified across multiple key quality parameters before release: DNA residue, GAG retention rate, and structural integrity testing
05
Custom Preservation & Delivery
Materials processed by lyophilization, irradiation sterilization, or wet preservation per partner requirements, balancing bioactivity and stability
Bioactive Preservation Comparison
| Dimension | Chemical (SDS) | HHP (Hopefan Biotech) |
|---|---|---|
| Bioactive Component Retention | ~20% (most active proteins chemically degraded) | ~91% (physical force, active components fully preserved) |
| ECM Ultrastructure | May be compromised (detergent disrupts fiber alignment) | Nanoscale preservation intact (verified by electron microscopy) |
| Chemical Residue Risk | Residual detergent present, requires additional wash validation | No chemical residues — pure physical processing |
| Decellularization Uniformity | Surface to moderate depth; limited efficacy in thick tissue | Uniform isostatic pressure; equally effective across deep thick tissue |
| Microbial Inactivation | Requires separate independent sterilization step | Inherent microbial inactivation capability |
| Processing Time | 24–72 hours | 15–60 minutes |
HHP retains approximately 4.5× more bioactive components than chemical methods, with no chemical residue risk.
Patent Protection
Patents & Core Technology
Built around ECM processing and structural preservation, the company has developed a comprehensive patent portfolio.
High-Pressure Decellularization Equipment (HHP)
一种医用材料处理用高压设备
Biological Tissue Processing Method for Transplantation
一种用于移植的生物组织处理方法
ECM Processing and Structural Preservation System
ECM Material Processing & Structural Preservation Technology
Technical Validation
The following metrics assess decellularization efficiency and structural preservation of ECM materials
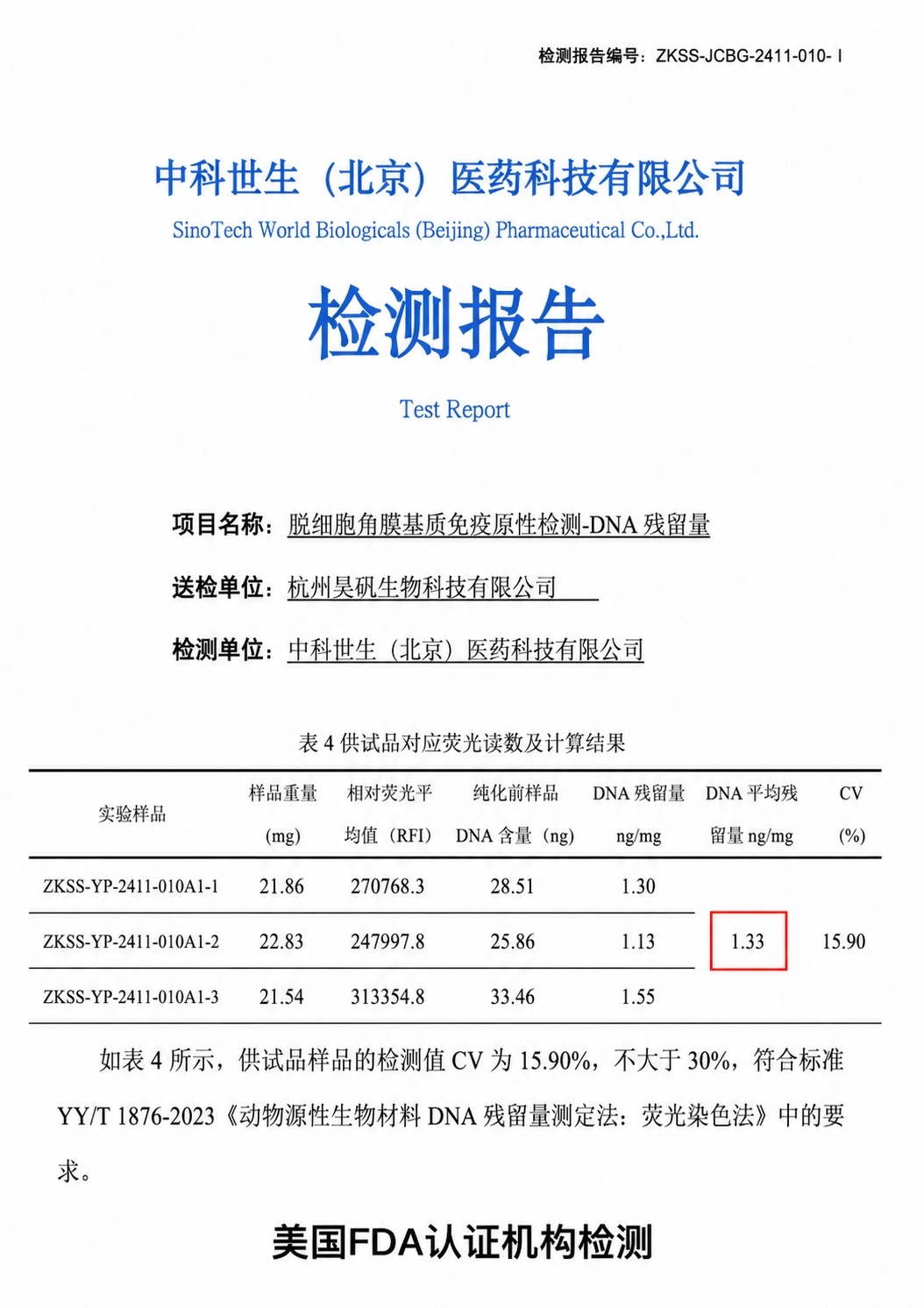
DNA Residue
Far below the industry reference threshold (<50 ng/mg)
Test results confirm DNA residue is far below the industry safety threshold, demonstrating consistent and thorough decellularization across batches.
Third-Party Test Report
Bioactivity Retention
91%
Chemical methods ~20%
Cell Removal Rate
>95%
GAG Retention
>99%
Nanostructure Preservation
Confirmed
Electron microscopy verified
Key quality indicators confirm that HHP technology achieves a stable balance between decellularization efficiency and structural preservation.
Technology Development
HHP decellularization method established at Japan's National Cerebral and Cardiovascular Center
HHP method refined and expanded at Tokyo Medical and Dental University
Product development and regulatory filing at Japan's National Institute of Biomaterials Engineering; joint R&D with listed Japanese corporation
Technology transferred to China; founding of HopeFan Biotech predecessor
Hopefan Biotech established; GMP cleanroom facility built; technology commercialized
View Product Catalog
DNA Residue: 1.33 ng/mg · Cell Removal Rate: >95%
